Wolfram|Alpha is a great tool for calculating the molarity, molality, mass fraction and amount fraction concentration of solutions. It also generates solution properties and preparation recipes.
Online Dilution Calculator: Chemistry Solvers
Solve dilution problems with Wolfram|Alpha
More than just an online dilution calculator
Learn more about:
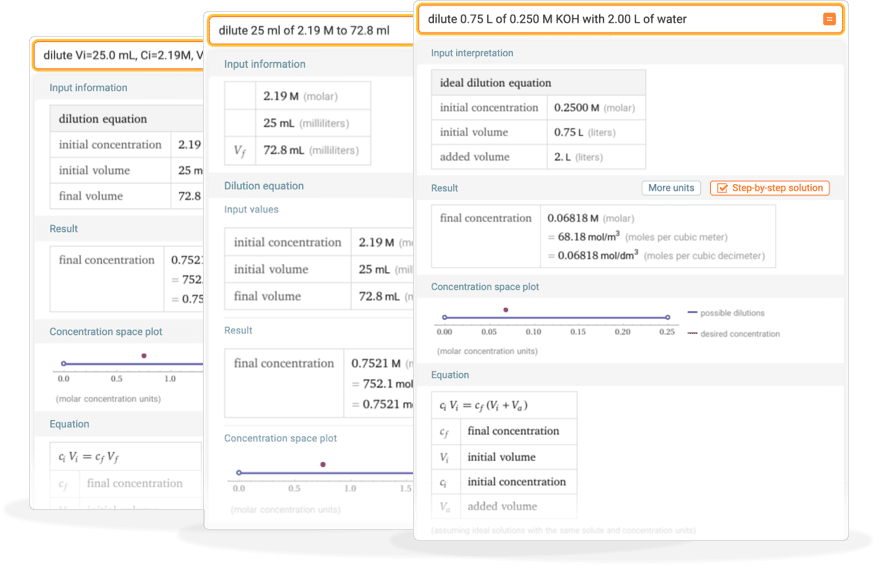
Tips for entering queries
Enter your queries using plain English. Here are some examples illustrating how to ask for a dilution.
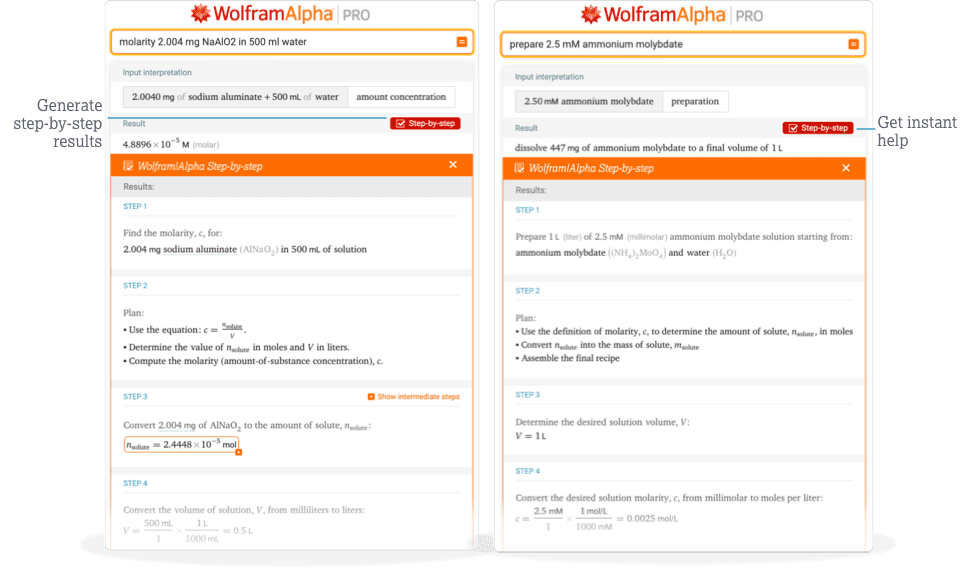
Access instant learning tools
Get immediate guidance with step-by-step support for chemistry.
Learn more about:
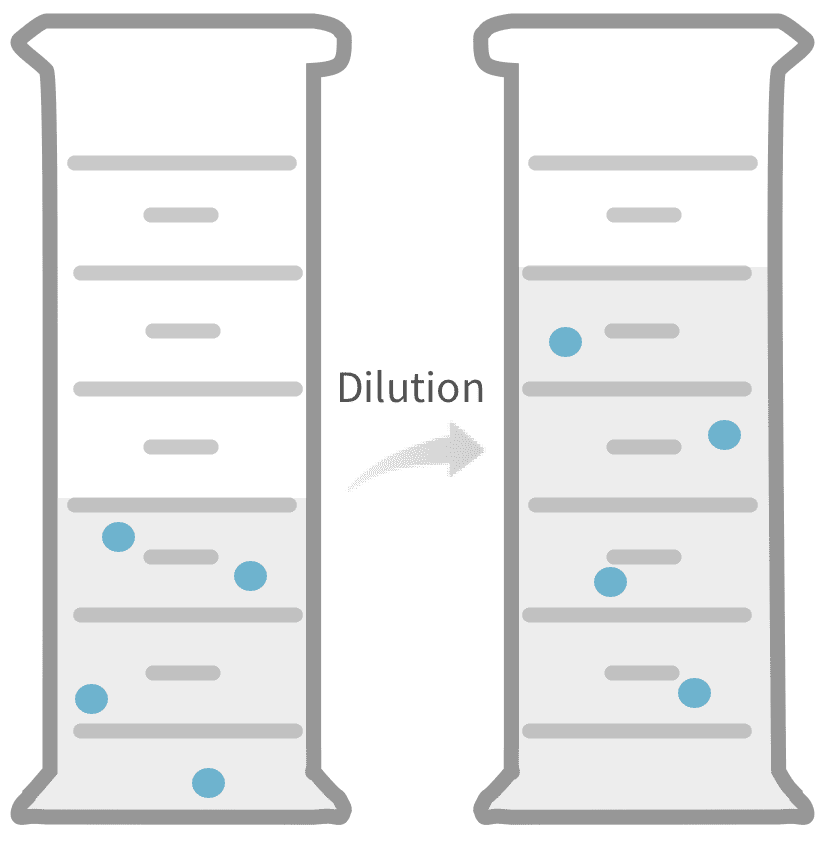
What are dilutions?
Dilution is an important experimental technique for creating solutions of a desired concentration.
A solution is a mixture of components that is homogenous at the molecular level. The solution components are two or more pure substances that were mixed to form the solution. For a binary solution containing two components, the minor component is called the solute and the other component is the solvent. The solute concentration describes the amount of solute dispersed in a given quantity of the solvent. Dilution is a process whereby the concentration of the solute in a stock solution is reduced by the addition of more solvent. Diluting a solution resulting from a previous dilution by adding the same amount of solvent is known as a serial dilution. Dilution calculations allow one to determine how much solvent to add or what the final concentration is after the addition of solvent.
How Wolfram|Alpha calculates dilutions
Wolfram|Alpha computes dilutions using the dilution equation, ciVi=cfVf, or the ideal dilution equation, ciVi=cfVi + Va, by calling FormulaData. The dilution equation is applicable for all dilutions since the non-additivity of solution volumes is accounted for. Solution volumes are not additive due to the intermolecular forces between the solute and solvent entities. If the intermolecular forces between the solute and the solvent are the same as the intermolecular forces in the pure solvent, the solution is said to be ideal and the ideal dilution equation can be used. In practice, the ideal dilution formula is only applicable for solutions of very low concentration or situations where errors in concentration do not affect experimental results.